Learn about our comprehensive antibody validation methods to ensure monospecificity. Antibody Validation>>

Formalin-fixed, paraffin-embedded human Placenta stained with Superoxide Dismutase 1 Mouse Monoclonal Antibody (SOD1/2089).
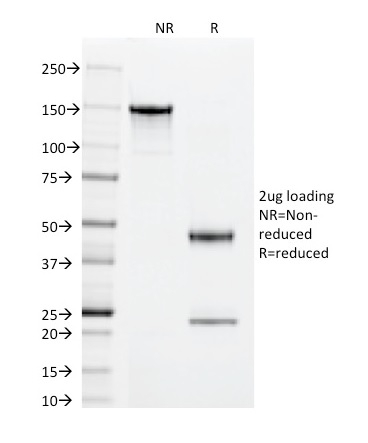
SDS-PAGE Analysis of Purified Superoxide Dismutase 1 Mouse Monoclonal Antibody (SOD1/2089). Confirmation of Integrity and Purity of Antibody.

Analysis of Protein Array containing more than 19,000 full-length human proteins using Superoxide Dismutase 1 Mouse Monoclonal Antibody (SOD1/2089). Z- and S- Score: The Z-score represents the strength of a signal that a monoclonal antibody (MAb) (in combination with a fluorescently-tagged anti-IgG secondary antibody) produces when binding to a particular protein on the HuProtTM array. Z-scores are described in units of standard deviations (SD's) above the mean value of all signals generated on that array. If targets on HuProtTM are arranged in descending order of the Z-score, the S-score is the difference (also in units of SD's) between the Z-score. S-score therefore represents the relative target specificity of a MAb to its intended target. A MAb is considered to specific to its intended target, if the MAb has an S-score of at least 2.5. For example, if a MAb binds to protein X with a Z-score of 43 and to protein Y with a Z-score of 14, then the S-score for the binding of that MAb to protein X is equal to 29.
Cu-Zn superoxide dismutase-1 (SOD-1) is a well-characterized cytosolic scavenger of oxygen free radicals that requires copper and zinc binding to potentiate its enzymatic activity. Enzymatically, SOD-1 facilitates the dismutation of oxygen radicals to hydrogen peroxide and also catalyzes pro-oxidant reactions, which include the peroxidase activity and hydroxyl radical generating activity. SOD-1 is ubiquitously expressed in somatic cells and functions as a homodimer. Defects in the gene encoding SOD-1 have been implicated in the progression of neurological diseases, including amyotrophic lateral sclerosis (ALS), a neurodegenerative disease characterized by the loss of spinal motor neurons, Down syndrome and Alzheimer s disease. In familial ALS, several mutations in SOD-1 predominate, resulting in the loss of zinc binding, the loss of scavenging activity of SOD-1, and correlate with an increase in neurotoxicity and motor neuron death.
There are no reviews yet.