
Formalin-fixed, paraffin-embedded human Placenta stained with PD-ECGF Recombinant Mouse Monoclonal Antibody (rTYMP/3444).
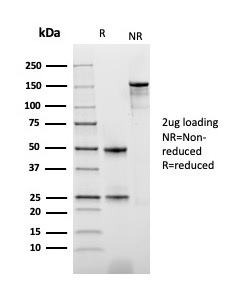
SDS-PAGE Analysis of Purified PD-ECGF Recombinant Mouse Monoclonal Antibody (rTYMP/3444). Confirmation of Integrity and Purity of Antibody
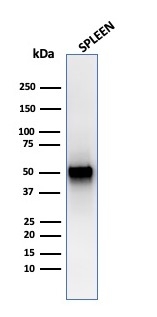
Western Blot Analysis of human spleen tissue lysate using PD-ECGF Recombinant Mouse Monoclonal Antibody (rTYMP/3444).
Recognizes a protein (amino acid 482) of 55kDa (in vivo 110kDa homodimer), identified as platelet-derived endothelial growth factor (PD-ECGF), same as thymidine phosphorylase (TP) or gliostatin. In the presence of inorganic orthophosphate, it catalyzes the reversible phospholytic cleavage of thymidine and deoxyuridine to their corresponding bases and 2-deoxyribose-1-phosphate. It is both chemotactic and mitogenic for endothelial cells and a non-heparin binding angiogenic factor present in platelets. Its enzymatic activity is crucial for angiogenic activity (metabolite is angiogenic). Higher levels of serum TP/PD-ECGF are observed in cancer patients. It is also involved in transformation of fluoropyrimidines, cytotoxic agents used in the treatment of a variety of malignancies, into active cytotoxic metabolites (e.g. 5′-deoxy-5-fluorouridine to 5-FU). High intra-cellular levels of TP/PD-ECGF are associated with increased chemosensitivity to such antimetabolites.
There are no reviews yet.