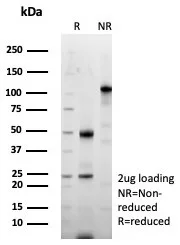
SDS-PAGE Analysis of Purified Periostin Recombinant Rabbit Monoclonal Antibody (POSTN/8523R). Confirmation of Purity and Integrity of Antibody.
Periostin (PN), also designated osteoblast-specific factor 2 (OSF-2), is a disulfide linked protein originally isolated as a osteoblast-specific factor. Periostin is a secreted protein that binds heparin and functions as a ligand for αVβ3 and αVβ5 integrins. In preosteoblasts, Periostin acts as a cell adhesion molecule and plays a role in osteoblast recruitment, spreading and attachment. Periostin is mainly detected in lower gastrointestinal tract, aorta, stomach, placenta, uterus and breast tissues but is up-regulated in epithelial ovarian tumors and overexpressed in breast cancer. Expression of Periostin is increased by bone morphogenetic protein (BMP2) and transforming growth factor β 1 (TGF β 1). Periostin contains a typical signal sequence, followed by a cysteine-rich domain, a fourfold repeated domain, which shows homology with the insect protein fasciclin, and a C-terminal domain.
There are no reviews yet.